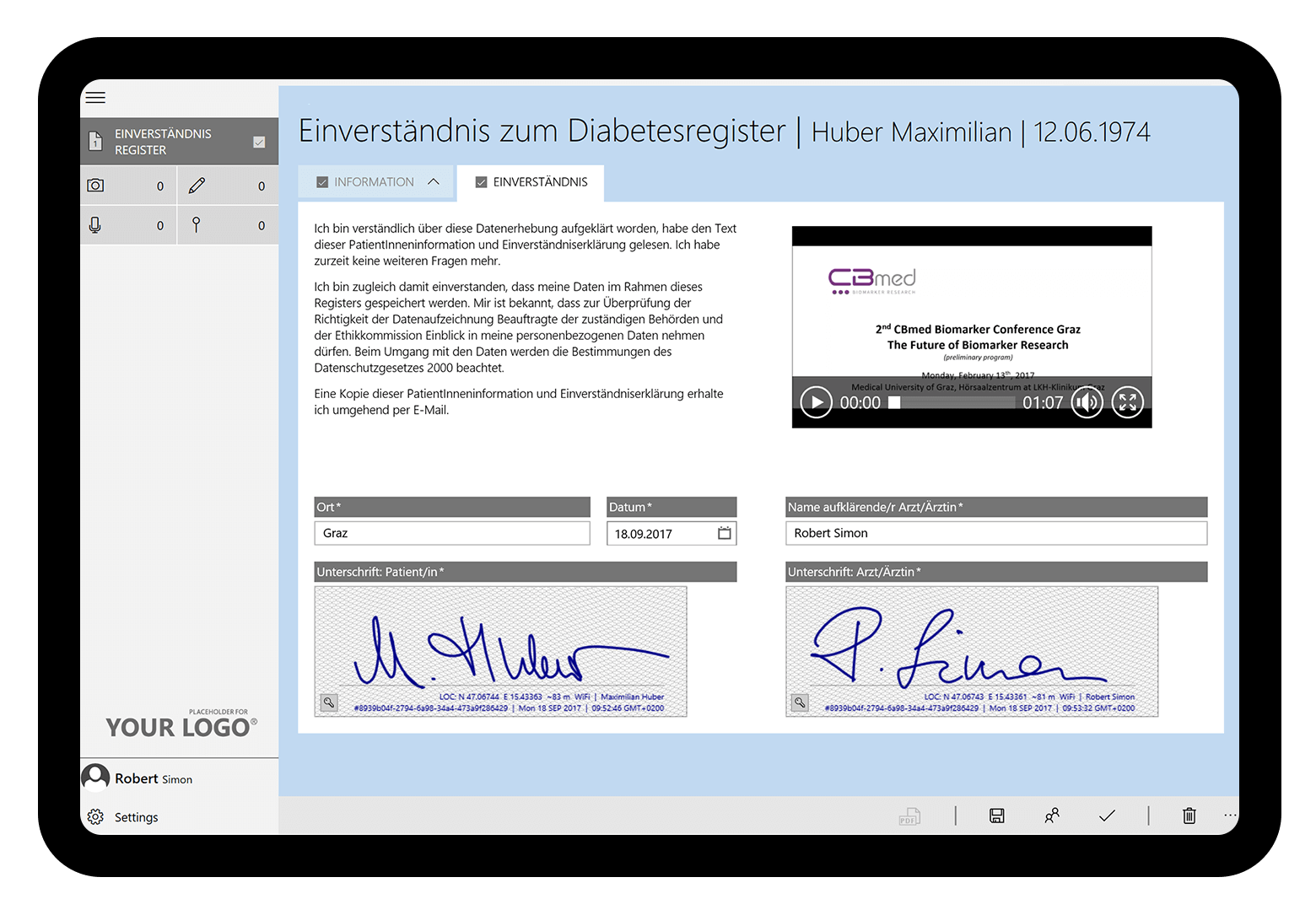
The Medical University of Graz captures patient data for diabetes research digitally and in a structured way with HybridForms. Data from patients’ written informed consent forms, results of medical visits, and protocols of medical examinations are captured digitally with tablets for the biomarker research diabetes registry.
HybridForms: Digital forms in medicine & research
The goal of the diabetes registry for biomarker research is to capture a representative cohort of patients routinely treated at the University Hospital Graz. Consents for participation in the clinical trials as well as data from individual medical visits and patient interviews are collected on tablets using the HybridForms mobile App.
All results are securely archived and transfered pseudonymized via interface to the clinical trial software. During pseudonymization, the patient’s name is replaced by a pseudonym (Study Subject ID) to protect the identity of individual patients. However, this procedure preserves the references between different data sets for further analysis.
Digitization makes all data available more quickly for further evaluations and scientific research – and the error rate is minimized thanks to transparent and consistently traceable documentation.

»The tablet-based data entry with HybridForms allows us to perform data collection in the Diabetes Biomarker Research Registry much faster, more efficiently and much more securely. I am convinced that we will come across this model very frequently in other future research projects.«

»As an implementation partner, HybridForms allows us to focus fully on our customers’ challenges – and thus achieve outstanding project results in short lead times.«
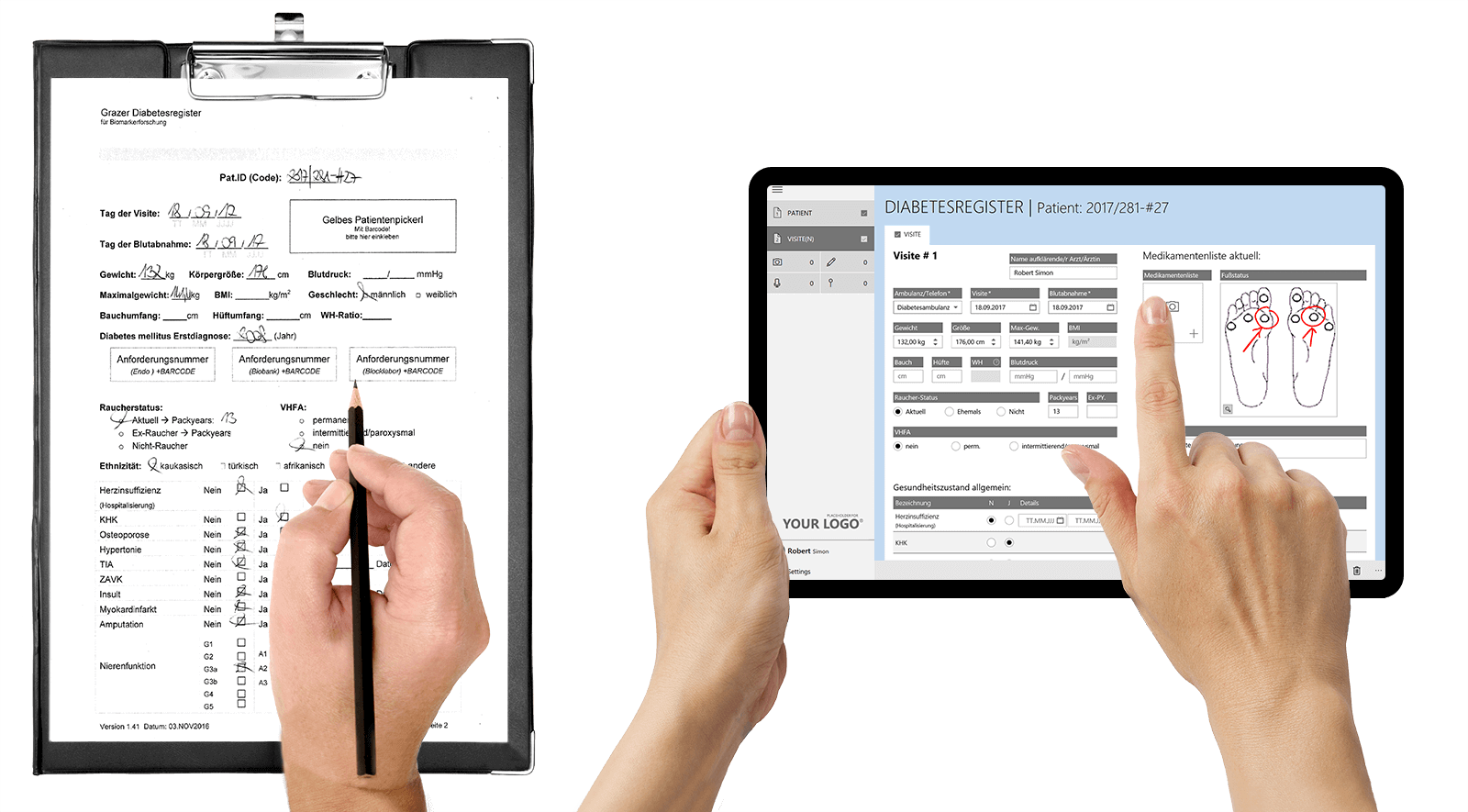
HybridForms App & Software with secure enterprise features: »Tablet instead of clipboard« for higher data quality, more transparency & more efficient clinical processes
Smart Health & Social Care
Administrative form-based processes are optimized through digitization with HybridForms, and healthcare professionals can work more efficiently with mobile & digital tools.
User-friendly pre-filled forms with integration of data from third-party systems (e.g. HIS) are processed via App on tablets and smartphones, and the entries are checked automatically. All data, photos and signatures are in one document – secure, traceable and legally valid.